MHS AP Chemistry
Young's Experiment
PART 1: Calibration for Young's Experiment
Observe and record the identity and color of the gas in the glowing
Geissler tube. These tubes contain gas at low pressure (about 0.001
atm). A high voltage electric discharge ionizes the gas, and the
recapture of the electrons to characteristic energy levels results in a
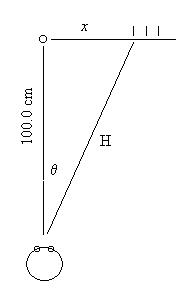
unique line spectrum. Place one meter stick parallel to the line
of sight and another behind the tube as diagrammed below.
Using diffraction glasses, observe the line spectrum of helium.
Twist your head until the colored lines are "standing" on the meter stick.
Your partner should stand behind the meter stick and use a pencil to help
you find the exact distance from the tube to each line above the meter
stick (marked x in the diagram). Record this distance in centimeters.
Use Table 1 below to determine the wavelength of each line (as
an alternative, you may use the spectroscope with the internal wavelength
scale and read it directly). Use the known wavelengths to determine
the diffraction grating width (d) as follows:
| The tube should be exactly 1.00 meters from your eyes.
Use 100.0 cm as L. |
|
L = 100.0 cm |
 |
| |
|
|
|
| The distance from the tube to the line is x. |
|
Record x for each line |
|
| |
|
|
|
| Calculate the distance from the diffraction grating to the line, using
the Pythagorean Theorem. This distance is the hypotenuse (H). |
|
L2 + x2
= H2 |
|
| |
|
|
|
| The spacing of diffraction grooves, angle of diffraction, and wavelegth
of the color line are related by the equation |
|
l = dsinq |
|
| |
|
|
|
| From trig, you may recall that |
|
|
|
| |
|
|
|
| Substitute and solve for d: |
|
|
|
|
|
|
|
| Calculate an average value for d. |
|
|
|
|
|
|
|
Calibration Data Table
(Helium)
[wavelengths
from CRC 83rd edition, p 10-28]
| Color |
l |
x |
H |
d |
|
(nm) |
(cm) |
(cm) |
(units: _____) |
|
|
|
|
|
| purple |
388.9* |
__________ |
__________ |
__________ |
| blue |
447.2 |
__________ |
__________ |
__________ |
| blue |
471.3* |
__________ |
__________ |
__________ |
| blue/green |
492.2* |
__________ |
__________ |
__________ |
| green |
501.6 |
__________ |
__________ |
__________ |
| yellow |
587.6 |
__________ |
__________ |
__________ |
| red |
667.8 |
__________ |
__________ |
__________ |
| red |
706.5* |
__________ |
__________ |
__________ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
PART 2: Application of Young's Experiment
Observe the line spectrum of hydrogen gas. Measure and record the distance
of each line from the glowing tube. Use the equations from PART 1 to determine
the wavelength of each line. From the wavelength, determine the associated frequency
and energy for each electron transition in excited hydrogen.
Once you have determined the energy of each line in the hydrogen spectrum,
use the Rydberg equation to determine from which energy levels the electrons
"fall" (find ni; nf
= 2).
Spectrum Data Table (Hydrogen)
| Color |
d |
x |
H |
l |
n |
E |
nf |
ni |
|
(_____) |
(cm) |
(cm) |
(_____) |
(_____) |
(_____) |
( -- ) |
( -- ) |
|
|
|
|
|
|
|
|
|
|
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
|
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
|
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
|
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
|
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
|
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
|
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
|
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
__________ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Table 1
Helium Spectrum
|
Table 2
Constants & Relations
|
Table 3
Colors & Information
|
| color |
l (nm) |
| purple |
388.9* |
| blue |
447.2 |
| blue |
471.3* |
| blue/green |
492.2* |
| green |
501.6 |
| yellow |
587.6 |
| red |
667.8 |
| red |
706.5* |
| * may be too faint to see. |
|
|
|
| h = 6.63x10-34 J/Hz |
| c = 3.00x108 m/s |
| c = ln |
| E = hn |
| RH = 2.18x10-18 J/photon |
| 1 m = 102 cm = 109
nm |
| DE
= hn = -RH |
é1 |
__ |
1ù |
|
| ënf2 |
|
ni2û |
|
|
|
Be very careful about the signs: What is the sign
for the frequency of light? What is the sign for the energy
change of the electron?
|
|
color |
wavelength (l) |
frequency (n) |
| |
[nm] |
[x1014Hz] |
|
violet |
400 to 420 |
7.5 to 7.1 |
|
blue |
420 to 490 |
7.1 to 6.1 |
|
green |
490 to 580 |
6.1 to 5.2 |
|
yellow |
580 to 590 |
5.2 to 5.1 |
|
orange |
590 to 650 |
5.1 to 4.6 |
|
red |
650 to 700 |
4.6 to 4.3 |
|
Attach one sheet with orderly calculations for d (be very careful about the
labels for d).
Attache one other sheet with orderly calculations for one of the values of
nf.
[Syllabus 6][MHS AP Chem page]
